How do isomers different from one another?
 Sophia Bowman
Sophia Bowman 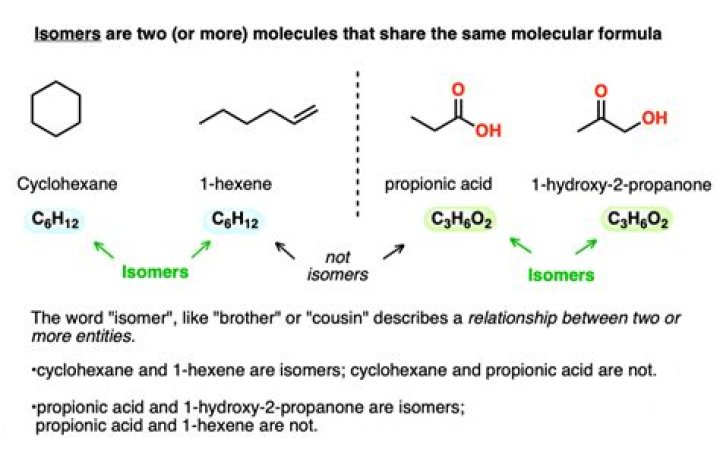
Isomers are compounds that contain exactly the same number of atoms, i.e., they have exactly the same empirical formula, but differ from each other by the way in which the atoms are arranged.
What is the main difference between the two types of isomers?
There are two broad types of isomers: constitutional and stereoisomers. Constitutional isomers differ in bonding and connectivity. Stereoisomers differ in 3D orientation. Enantiomers and diastereomers are two sub-types of stereoisomers.
What are structural isomers of each other?
In chemistry, a structural isomer (or constitutional isomer in the IUPAC nomenclature) of a compound is another compound whose molecule has the same number of atoms of each element, but with logically distinct bonds between them. The term metamer was formerly used for the same concept.
What are the 3 types of isomers?
There are three types of structural isomers: chain isomers, functional group isomers and positional isomers. Chain isomers have the same molecular formula but different arrangements or branches. Functional group isomers have the same formula but different functional groups.
Can structural isomers have double bonds?
Notice that in the structural isomers, there was some difference in the connection of atoms. For example, 1-butene has a double bond followed by two single bonds while 2-butene has a single bond, then a double bond, then a single bond. A stereoisomer will have the same connectivity among all atoms in the molecule.
What are the two types of isomers?
There are two general types of isomers. Constitutional isomers are molecules of different connectivity—analogous to simple bracelets in which the order of red and green beads is different. The second type is stereoisomers. In stereoisomers the connectivity is the same, but the parts are oriented differently in space.
What are the 3 main types of isomers?
What are structural isomers give examples?
Structural isomers are molecules with the same molecular formula, but their atoms have different arrangements or bonds. for example : Butane and isobutane have the same number of carbon (C) atoms and hydrogen (H) atoms, so their molecular formulas are the same.
How many types of structural isomers are there?
There are three types of Structural isomerism existing namely chain isomerism, position isomerism and functional group isomerism.
What is structural isomers give examples?
What’s the difference between stereoisomers and structural isomers?
The key difference between structural isomers and stereoisomers is that structural isomers have the same chemical formula, but different atomic arrangements, whereas stereoisomers have the same chemical formula and atomic arrangement, but different spatial arrangements.
Which is an example of functional group isomerism?
Functional group isomerism – structural isomers with the same molecular formula, but with atoms connected differently so dissimilar functional groups are formed. Butane and isobutane (C 4 H 10) are structural isomers of each other. Pentan-1-ol, pentan-2-ol, and pentan-3-ol are structural isomers that exhibit position isomerism.
How are geometric isomers different from optical isomers?
Moreover, there are two groups of stereoisomers as geometric isomers and optical isomers. Geometric isomers are what we call cis-trans isomers. There are always two isomers as cis isomer and trans isomer. Therefore, they always occur as a pair. Moreover, an organic compound must have double bonds in order to have geometric isomers.
How are cis isomers different from trans isomers?
There are always two isomers as cis isomer and trans isomer. Therefore, they always occur as a pair. Moreover, an organic compound must have double bonds in order to have geometric isomers. Here, the isomers are different from each other according to the attachment of functional groups to the vinyl carbon atoms (carbon atoms in the double bond).



