How many p orbitals are occupied in an atom?
 Sophia Bowman
Sophia Bowman 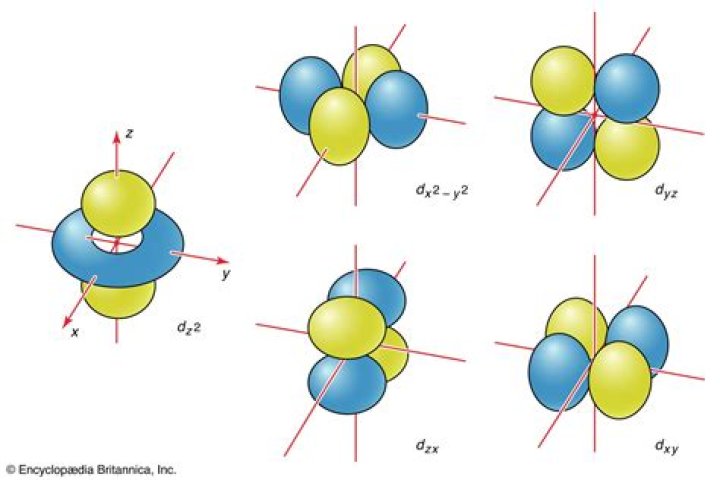
3 orbitals
The p sublevel has 3 orbitals, so can contain 6 electrons max. The d sublevel has 5 orbitals, so can contain 10 electrons max.
How do you find occupied p orbitals?
As you can see, the 2p and 3p sublevels each hold six electrons, which means that they are completely occupied. Since every p sublevel has a total of three p-orbitals – px , py , and pz – the number of p-orbitals occupied in a K atom is equal to 6 – 3 p-orbitals on the 2p sublevel and 3 p-orbitals on the 3p sublevel.
How many electrons occupy p orbitals?
6 electrons
Explanation: There are three types of p-orbital and each orbital can hold two electrons, thus 6 electrons can occupy the ‘p-orbitals’ before breaking Pauli’s Exclusion principle.
How many p orbitals are occupied in a oxygen atom?
there’s one is the “s” shell, 3 in the “p” shell, 5 in the “d” shell and 7 in the “f” shell. So if the electron configuration for oxygen(O) is 1s^2,2s^2,2p^4/(He)2s^2,2p^4, then there are 5 total orbitals, 1s,2s and 2p.
Which orbitals have the highest energy?
In all the chemistry of the transition elements, the 4s orbital behaves as the outermost, highest energy orbital.
How many p orbitals are there in NA?
Since 1s can only hold two electrons the next 2 electrons for sodium go in the 2s orbital. The nex six electrons will go in the 2p orbital. The p orbital can hold up to six electrons. We’ll put six in the 2p orbital and then put the remaining electron in the 3s.
Why are there 3 p orbitals?
Not only hydrogen atom, but in all the atoms there are 3 p orbitals in any energy state because p orbital has azimuthal quantum number 1, therefore it has 3 orbitals px,py and pz with magnetic quantum numbers -1,0,1.
What does P orbital look like?
A p orbital is shaped like 2 identical balloons tied together at the nucleus. The orbital shows where there is a 95% chance of finding a particular electron. The p orbitals at the second energy level are called 2px, 2py and 2pz. There are similar orbitals at subsequent levels: 3px, 3py, 3pz, 4px, 4py, 4pz and so on.
Why does p orbitals have 6 electrons?
The 2p, 3p, 4p, etc., can each hold six electrons because they each have three orbitals, that can hold two electrons each (3*2=6).
Does hydrogen have p electrons?
All the 2p orbitals have exactly the same energy. Hydrogen only has one electron and that will go into the orbital with the lowest energy – the 1s orbital. This is because the p orbitals all have the same energy and the electrons prefer to be on their own if that’s the case.
How many orbitals are in oxygen?
Oxygen has the electron configuration 1s22s22p4, with two unpaired electrons (one in each of the two 2p orbitals). Valence bond theory would predict that the two O–H bonds form from the overlap of these two 2p orbitals with the 1s orbitals of the hydrogen atoms.
Why is 3rd shell 8 or 18?
Originally Answered: According to the 2-8-8 rule, the 3rd orbital has only 8 electrons in its valence orbital, but from the formula 2n^2, the 3rd shell would have 18 electrons in its valence orbital.
Is 4s higher than 3d?
The 4s electrons are lost first followed by one of the 3d electrons. The electrons lost first will come from the highest energy level, furthest from the influence of the nucleus. So the 4s orbital must have a higher energy than the 3d orbitals.
What are the 4 types of orbitals?
Named for their energy sublevels, there are four types of orbitals: s, p, d, and f. Each orbital type has a unique shape based on the energy of its electrons.
What are the 3 p orbitals?
For any atom, there are three 3p orbitals. These orbitals have the same shape but are aligned differently in space. The three 3p orbitals normally used are labelled 3px, 3py, and 3pz since the functions are “aligned” along the x, y, and z axes respectively. Each 3p orbital has four lobes.
Why is it called p orbital?
You might expect that the ‘s’ stands for ‘spherical’ and ‘p’ stands for ‘polar’ because these imply the shapes of the s and p orbitals, but unfortunately, the letter designations have nothing to do with the orbital shapes.
What is the meaning of P in p orbital?
: the orbital of an electron shell in an atom in which the electrons have the second lowest energy.
What is 1s 2s 2p 3s 3p?
1s 2s 2p 3s 3p represents the electron orbital energy levels.
Why does P have 3 orbitals?
Are electron orbitals real?
The term ‘orbital’, though, is frequently used with a different meaning. With Page 8 Are orbitals observable? 31 each one-electron wave function comes a probability density function on real space for the electron’s position. When it carries this meaning, therefore, orbitals are experimental quantities.
How many p orbitals are filled?
three p orbitals
This makes a total of three p orbitals to be filled in any given energy level from the second up. Each of these three p orbitals needs to be filled with electrons in the second and higher energy levels. But there is another rule to follow in placing the electrons in the p orbitals. This is Hund’s Rule.
Which orbitals are p orbitals?
p orbitals At the first energy level, the only orbital available to electrons is the 1s orbital, but at the second level, as well as a 2s orbital, there are 2p orbitals. A p orbital is shaped like 2 identical balloons tied together at the nucleus.
How many p orbitals are in oxygen?
three 2p orbitals
Oxygen has three 2p orbitals. A maximum of six electrons can be retained by the p subshell since there are three orbitals inside this sub shell. At right angles to each other, the three p orbitals are and have a lobed shape.
Which electron orbital has the highest energy?
- The larger the number of the energy level, the farther it is from the nucleus.
- Each subshell has a maximum number of electrons which it can hold: s – 2 electrons, p – 6 electrons, d – 10 electrons, and f – 14 electrons.
- Usually mid orbitals has highest energy.
Why do p orbitals have 6 electrons?
The p orbital is a dumbbell-shaped or lobed region describing where an electron can be found, within a certain degree of probability. The shape of the orbital depends on the quantum numbers associated with an energy state.
How many electrons do you get in a p orbital?
Therefore, the number of electrons you get in p-orbitals will be equal to #11# 6 electrons in the 2p-subshell 5 electrons in the 3p-subshell
How many electrons are occupied in a K atom?
Potassium ( K) is located in group 1, period 4 of the periodic table and has an atomic number of 19. Since you’re dealing with a neutral atom, the number of electrons K has must equal 19. As you can see, the 2p and 3p sublevels each hold six electrons, which means that they are completely occupied.
How many electrons occupy a p orbital in a chlorine atom?
Now, notice that the first energy level doesn’t not contain a p-subshell, and implicitly any p-orbitals. The second energy level, on the other hand, contains two subshells, the 2s-subshell, which contains 2 electrons, and the 2p-subshell, which contains three 2p-orbitals and a total of 6 electrons, 2 per orbital. Finally,…